
Mexico and Argentina will produce the AstraZeneca COVID-19 vaccine
The vaccine developed by Oxford University and AstraZeneca laboratories will be distributed across Latin America.
The governments of Mexico and Argentina have confirmed that their countries will be producing between 150 and 250 million doses of a promising COVID-19 vaccine.
The vaccine was developed by Oxford University and AstraZeneca, the United Kingdom’s second largest pharmaceutical company.
The millions of doses are expected to be delivered equitably across Latin America early next year.
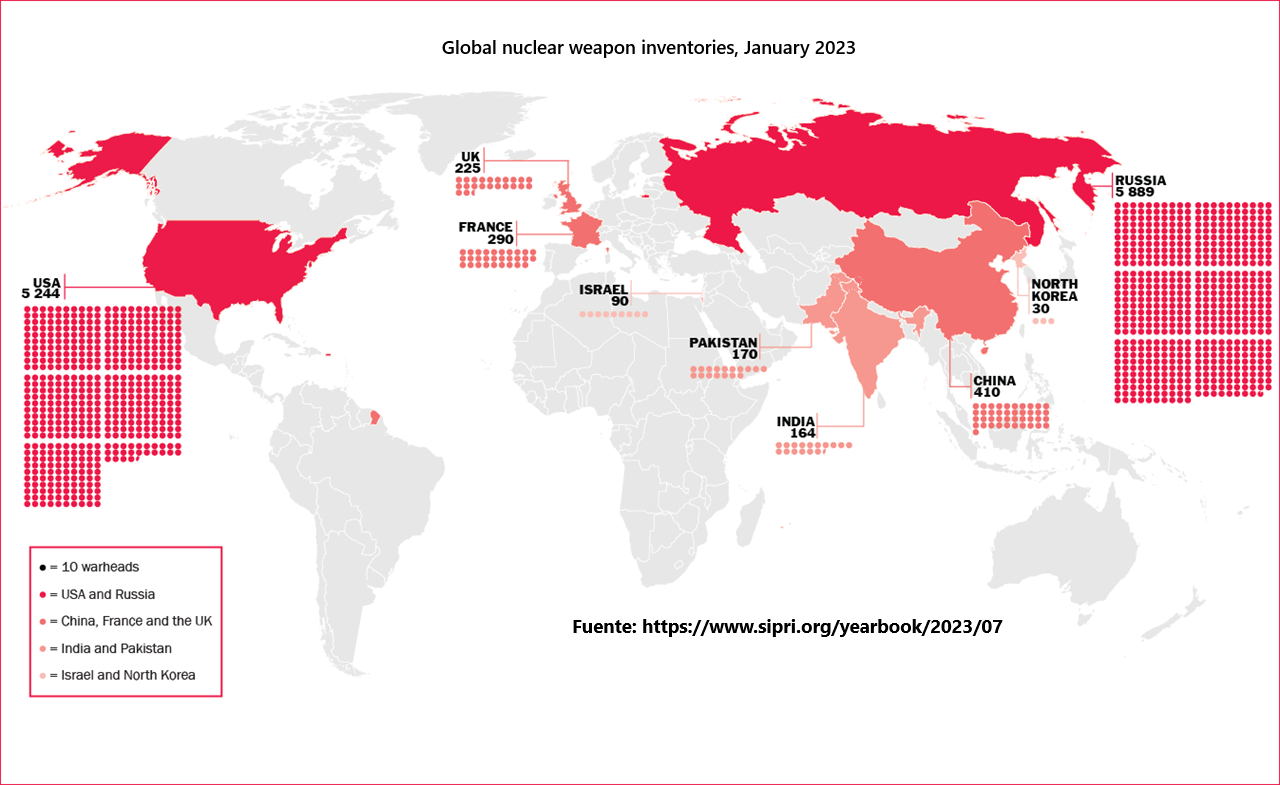
Latin America has become an epicenter for the novel coronavirus with the region having over 5.8 million cases and losing close to 230,000 lives to the disease.
Argentinian president Alberto Fernández disclosed more information on the agreement in a meeting on Wednesday.
“The laboratory has signed an agreement with the Slim Foundation to produce between 150 and 250 million vaccines for all of Latin America except Brazil,” he said.
The Carlos Slim Foundation is helping countries across the region obtain doses of the vaccine at a subsidized cost. It is estimated that the price per dose will be between three and four dollars.
Brazil will be the only country in the region to not receive doses from the production. It signed an independent accord with Oxford University to acquire 100 million doses as well as the technology to be able to produce the vaccine in their own laboratories.
Brazil is one of the countries that has been most impacted by the novel coronavirus. They trail only the U.S. in both cases and deaths related to the virus. They have the fifth-highest deaths per 100,000 residents which further shows how their healthcare system has been overwhelmed in recent months.
This has forced state governments within the country to seek other promising vaccines aside from the one developed by AstraZeneca.
São Paulo has allowed Chinese firm Sinovac and U.S. pharmaceutical giant Pfizer to each conduct thousands of trials of their vaccines in Brazil’s most populated state.
The neighboring state of Paraná reached an agreement on Wednesday to produce Russia’s vaccine, which is named Sputnik V as a tribute to the world’s first satellite launched into space by the Soviet Union.
Russian president Vladimir Putin announced that his country’s vaccine is the first in the world to give regulatory approval to a COVID-19 vaccine and that they will start a mass immunization program in October.
RELATED CONTENT
“As far as I know, this morning for the first time in the world a vaccine against the novel coronavirus infection was registered. I know this very well because one of my daughters has been vaccinated with such a vaccine,” Putin said.
Russia is racing to be recognized as the country that solved the coronavirus pandemic, but the lack of transparency from their scientists when developing Sputnik V leaves many western officials with serious doubts.
“Having a vaccine and proving that a vaccine is safe and effective are two different things. We have half a dozen or more vaccines, so if we wanted to take the chance of hurting a lot people or giving them something that doesn’t work we could start doing this next week if we wanted to but that’s not the way it works,” said Anthony Fauci, America’s top infectious disease expert.
The World Health Organization has urged Russia to not massively produce the vaccine until they have completed “Phase 3” of testing, but the Brazilian state of Paraná and the Philippines are eager to start large-scale trials in the Fall.
Mexican president Andrés Manuel López Obrador talked about the vaccine his country will soon be producing at his morning press conference on Thursday.
He also reassured the Mexican public that they would not have to pay for the treatment and that it will be universal.
“We have to fulfill what is established in article 4 of the constitution, the people's right to health. This is free medical care, medicine and vaccines... all Mexicans will have access to the vaccine and there should be no concern for poor people,” said AMLO.
The University of Oxford confirmed last month that their vaccine is safe to test on humans and that results did see a production of antibodies needed to develop immunity from the virus.
The risk is that although the AstraZeneca vaccine has been through two phases of tests, there is no guarantee that it will be effective on all recipients.









LEAVE A COMMENT: