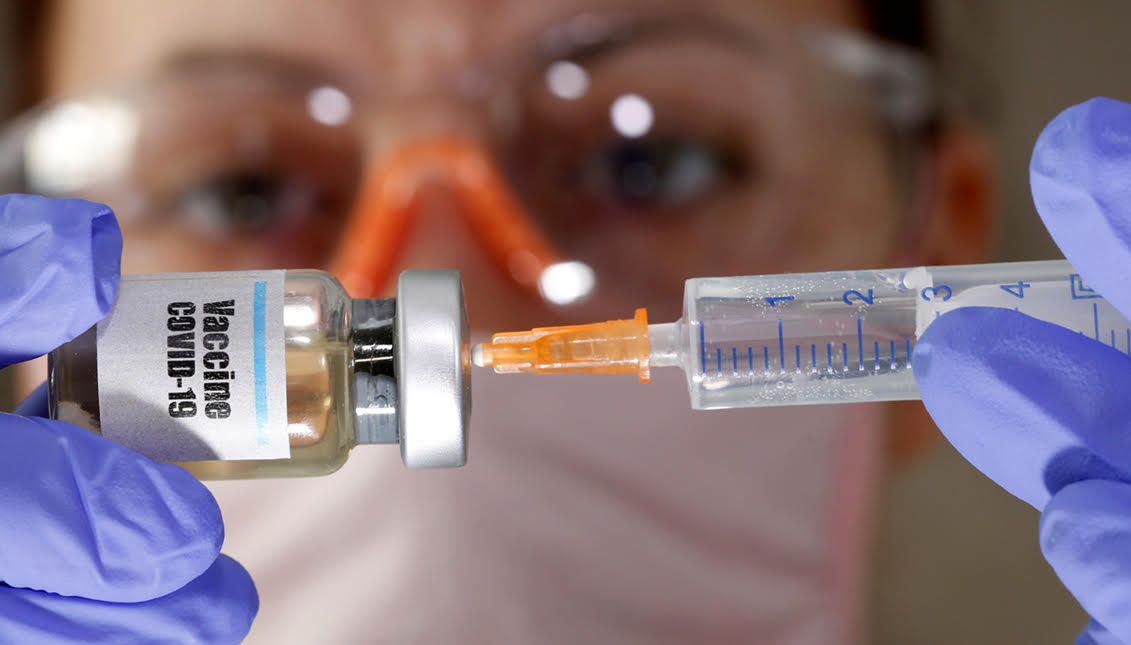
Moderna files for vaccine approval amid soaring Covid-19 cases
Fast-track vaccines are on the horizon, but the coronavirus positivity rates are spiking.
The year 2020 has felt like an entire decade packed into 11 months. Now, at the end of November and beginning of December, Covid-19 cases are at an all-time high, and the “curve” looks much more like a scaling mountain during this second wave with no real shutdowns.
There is, however, a solution, with multiple vaccines on the way.
The partnership of Pfizer and BioNTech were the first to fast track a vaccine on Nov. 20, and now Moderna, another pharmaceutical giant, will apply for authorization of its own Covid-19 vaccine on Nov. 30 from the U.S. Food and Drug Administration (FDA).
Clinical trials showed that the vaccine is 94% effective at preventing people from getting Covid-19, and 100% effective at preventing a severe case of the virus. Despite the recorded success, there are still people who are doubting the vaccine, and even saying they won’t get it after it becomes widely available.
Before Pfizer and Moderna released promising vaccine news, a Gallup poll found 42% of U.S. adults said they would *not* get a COVID vaccine when it’s first available.
— The Recount (@therecount) November 30, 2020
If you’re hesitant about receiving the vaccine, tell us about your biggest questions or concerns.
“We have a full expectation to change the course of this pandemic,” said Moderna’s Chief Medical Officer, Dr. Tal Zaks, after he saw the data of the vaccine on Nov. 28.
RELATED CONTENT
Working quickly as the pandemic spread around the globe, Moderna gave its vaccine to about 30,000 people in the United States, with some also getting a placebo shot. Months after the initial shot of the vaccine, of the 15,000 people who actually received it, only 11 people actually contracted the virus.
Now that testing is done and the applications have been filled out, both Pfizer and Moderna have meetings scheduled with the Vaccines and Related Biological Products Advisory Committee on Dec. 10 and Dec. 17.
As for the distribution? Frontline workers will be the first to receive the shots. Both vaccines are given in two doses that are weeks apart. Like any other vaccine or medication, there are side effects. This includes body aches for a couple days and potentially a fever.
With one month left of 2020, Moderna expects to have about 20 million doses available for the U.S., and 500 million to 1 billion doses globally for next year, according to a company press release.
Until then, the country will continue to see soaring cases, so wear a mask, wash your hands, keep gatherings as small as possible, and try to remain optimistic. There is a light at the end of the tunnel.










LEAVE A COMMENT:
Join the discussion! Leave a comment.